Dermatology
Abstract
Authors
Natalie E. Andresen, Heehwa G. Son, Joongho J. Joh, Shadmehr Demehri
Abstract
Enhancing NAD+ levels with nicotinamide riboside (NR) confers anti-inflammatory effects in human disease, although immunoregulatory mechanisms remain poorly characterized. We previously showed that ex vivo NR supplementation of primary CD4+ T cells from psoriatic individuals dampened immune responsiveness. To validate this in vivo, we performed a randomized, placebo-controlled NR supplementation study in individuals with mild-to-moderate psoriasis. Participants received oral NR (500 mg twice daily) or matching placebo for 4 weeks, with blood samples collected at baseline and after supplementation. NR reduced Th17 immune responsiveness. Bulk CD4+ T cell RNA-seq identified induction of the SLIT-ROBO signaling pathway. NR supplementation increased circulating SLIT2 levels and enhanced SLIT2 production in dermal fibroblasts. Pharmacologic and genetic interrogation in CD4+ T cells and fibroblasts demonstrated that SLIT2, acting through the ROBO1 receptor, inhibited Rho GTPase signaling, thereby attenuating canonical Th17 polarization and fibroblast inflammatory activation. These findings indicate that NAD+ augmentation exerts anti-inflammatory effects in psoriasis through SLIT2-ROBO1-mediated crosstalk between dermal fibroblasts and circulating CD4+ T cells, leading to suppression of Th17-driven inflammation.
Authors
Kim Han, Rachael J. Klein, Thomas C. Recupero, Anna Chiara Russo, Rahul Sharma, Anand K. Gupta, Shahin Hassanzadeh, Rebecca D. Huffstutler, Pradeep K. Dagur, Bryan Fisk, Neelam R. Redekar, Michael N. Sack
Abstract
Impaired adhesion and differentiation of keratinocytes is a hallmark of several skin diseases, but only some of the factors that regulate these processes have been identified. Here, we studied the role of isoform-rich dermokine – a wound- and tumor-regulated protein – in keratinocytes using a combination of multi-omics and functional approaches. CRISPR/Cas9-induced knockout of dermokine isoforms in human keratinocytes inhibited differentiation of these cells in three-dimensional organotypic skin cultures, which was confirmed by quantitative proteomics. In two-dimensional monocultures, dermokine deficiency affected the proteome and phosphoproteome as revealed by mass spectrometry. We found reduced abundance of differentiation-specific proteins and increased phosphorylation of cell adhesion protein p120 (catenin-δ1). The adhesive strength of dermokine knockout keratinocytes was impaired, which was rescued by p120 knock-down or ROCK inhibition. Finally, we verified the correlation between decreased dermokine expression and increased p120 phosphorylation in human non-healing wounds. These results identify dermokine as regulator of keratinocyte adhesion and differentiation, involving at least in part its effect on p120 phosphorylation and ROCK. Our data point to a function of dermokine in the pathogenesis of chronic wounds.
Authors
Vahap Canbay, Till Wüstemann, Weihua Tian, Tobias A. Beyer, Camilla Reiter Elbæk, Michael Stumpe, Gaetana Restivo, Chatpakorn Christiansen, Anabel Migenda Herranz, Susanne Mailand, Jürg Hafner, Rune Busk Damgaard, Steffen Goletz, Jörn Dengjel, Ulrich auf dem Keller, Chiara Francavilla
Abstract
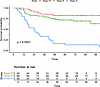
Merkel cell carcinoma (MCC) is a neuroendocrine carcinoma of the skin characterized by poor prognosis. This study aimed to explore the relationship between genetic alterations, tumor mutational burden (TMB), and MCC-specific survival (MCC-SS) in patients who underwent genomic profiling of tumors with OncoPanel. Univariate and multivariable analysis were used to assess the impact of genetic alterations on MCC-SS. Of the 188 identified patients, 164 were included in the analysis. The cohort had a mean age of 72.4 years (SD = 11.03), including 61.6% male. The median TMB was 5.32 (IQR = 3.04–25.53). Kaplan-Meier curves by high versus low TMB were significantly different (log-rank test, P = 0.017). PIK3CA (adjusted P = 0.003), SETBP1 (adjusted P = 0.002), KDR (adjusted P = 0.028), and RET (adjusted P = 0.033) were selected for multivariable analysis. In the multivariable regressions, only PIK3CA (HR = 2.07 [95% CI, 1.10–3.88]; P = 0.024) remained significant. PIK3CA remained significant across prespecified sensitivity analyses. In this study, high TMB and PIK3CA alterations were associated with poor MCC-SS. Identifying a higher-risk subgroup may inform risk stratification and motivate further evaluation of PI3K pathway targeting in future studies.
Authors
Matheus Lobo, Furkan Bahar, Julia L. Schnabel, Joao P. Duprat Neto, Aniket Shetty, Karam Khaddour, Manisha Thakuria, Ann W. Silk, James A. DeCaprio
Abstract
Prurigo nodularis (PN) is a chronic inflammatory skin disease characterized by pruritic skin nodules of unknown etiology. Little is known about genetic changes in PN pathogenesis, particularly somatic events, which are often implicated in inflammatory conditions. We thus performed whole-exome sequencing on 54 lesional and nonlesional skin biopsies from 17 patients with PN and 10 patients with atopic dermatitis (AD) for comparison. Somatic mutational analysis revealed that PN lesional skin harbors recurrent somatic mutations in fibrotic, neurotropic, and cancer-associated genes that are absent in adjacent PN nonlesional skin. Nonsynonymous mutations were most frequently present in NOTCH1 and the Notch signaling pathway, a key regulator of cellular proliferation and tissue fibrosis. In contrast, NOTCH1 mutations were absent in AD. Somatic copy-number analysis, combined with expression data, identified recurrently deleted and downregulated genes in PN lesional skin, which are associated with axonal guidance and extension. Follow-up immunofluorescence validation demonstrated increased NOTCH1 expression in PN lesional skin fibroblasts and increased Notch signaling in PN lesional dermis. Finally, a multicenter analysis revealed increased risk of NOTCH1-associated diseases in patients with PN. In characterizing the somatic landscape of PN, this study highlights the potential role of Notch pathway dysregulation in PN pathogenesis and fibrosis.
Authors
Ahmad Rajeh, Shahin Shahsavari, Hannah Cornman, Alexander Kollhoff, Anuj Gupta, Mindy D. Szeto, Anusha Kambala, Olusola O. Oladipo, Varsha Parthasarathy, Junwen Deng, Melika Marani, Shirin Shahsavari, Selina M. Yossef, Vedha Vaddaraju, Waleed Adawi, Yagiz M. Akiska, Davies M. Gage, Sarah Wheelan, Thomas Pritchard, Madan M. Kwatra, Yevgeniy R. Semenov, Alexander Gusev, Won Jin Ho, Srinivasan Yegnasubramanian, Shawn G. Kwatra
Abstract
Authors
Xiangmei Hua, Pranali N. Shah, Gustavo A. Velasquez, Lillian Sidky, George A. Romar, Lydia W. Boer, Natalie Hickerson, Tracy Qiying Cui, Federico Repetto, Abigail Waldman, Marilyn G. Liang, J. Paul Marcoux, MacLean Sellars, Victor Barrera, Birgitta A.R. Schmidt, Arash Mostaghimi, Ruth K. Foreman, Christine G. Lian, Sherrie J. Divito
Abstract
Authors
William J. Crisler, Noor Sohail, Samuel J. Steuart, Maria Vazquez-Machado, Arjun Mahajan, Maureen Whittelsey, Alex Pickering, Michael J. Martinez, Theresa Hutchins, Jessica E. Teague, Qian Zhan, Shannan Ho Sui, Ruth Ann Vleugels, Kathryn S. Torok, Heidi Jacobe, Rachael A. Clark, Avery LaChance
Abstract
Cutaneous radiation injury is an unintended consequence of radiotherapy for many common cancers and can progress to debilitating radiation-induced skin fibrosis (RISF). Existing radiation injury models do not fully capture the skin toxicities observed in patients, contributing to the lack of efficacious therapies to mitigate RISF. To address this, we developed an ex vivo human skin model that recapitulates the temporal radiation injury and RISF response. Human skin explants (N=12) subjected to ionizing radiation demonstrated DNA double-strand breaks and robust p53-driven transcriptional programming of cell cycle arrest, apoptosis, and senescence compared to non-irradiated controls. Irradiated skin also exhibited induction of pro-inflammatory cytokines, epithelial-mesenchymal transition, pro-fibrotic TGF-beta1 (TGFB1)-mediated signaling, and thickened collagen over time. P53 regulators murine double minute 2 (MDM2) and microRNA (miR)-34a were induced post-irradiation and may be leveraged to modulate injury response. Notably, RNA-sequencing of breast skin from mastectomy patients post-radiotherapy showed similar p53, inflammatory, and TGFB1 signatures as the ex vivo model, supporting its translational relevance. Together, this model provides a platform for identifying biomarkers and testing therapies to prevent or mitigate cutaneous radiation toxicities. Targeting the dynamic p53-driven pro-fibrotic radiation response represents a new therapeutic avenue to improve post-radiotherapy quality of life for cancer survivors.
Authors
Caroline Dodson, Sophie M. Bilik, Gabrielle DiBartolomeo, Hannah Pachalis, Lindsey G. Siegfried, Jordan A. K. Johnson, Seth R. Thaller, Irena Pastar, Marjana Tomic-Canic, Anthony J. Griswold, Rivka C. Stone
Abstract
This study provides a comprehensive evaluation of the cutaneous adipose tissue (CAT) transcriptome in psoriasis patients and investigates the effects of IL-17 blockade on CAT inflammation through a randomized placebo-controlled trial using secukinumab (ObePso-S study, NCT03055494). RNA sequencing analysis of CAT biopsies from 82 psoriasis patients revealed 2132 differentially expressed transcripts compared to healthy controls. Notably, significant gene dysregulation was observed in both lesional skin (LS)- and non-lesional (NL)-CAT, including activation of IL-17-driven pathways, antimicrobial peptide-related, and neutrophil degranulation signatures. Stratification by obesity demonstrated that obese psoriatic CAT exhibited more than 2-fold higher number of differentially expressed genes than non-obese counterparts, suggesting a synergistic interaction between psoriasis and obesity in driving CAT inflammation. Treatment with secukinumab markedly improved inflammatory signatures in psoriatic CAT, with greater improvements observed in obese patients. These findings reveal a pronounced and partially dependent on IL-17-dependent inflammatory phenotype in psoriatic CAT, challenge the conventional concept of psoriasis as a solely superficial skin disease, and highlight CAT as an important contributor to systemic inflammation in psoriasis.
Authors
Naomi Shishido-Takahashi, Sandra Garcet, Inna Cueto, Hong Beom Hur, Elisa F. Muscianisi, Jennifer Steadman, Andrew Blauvelt, James G. Krueger
Abstract
Dermal fibrosis is a cardinal feature of systemic sclerosis (SSc) for which there are limited Systemic sclerosis (SSc) is characterized by dermal fibrosis accompanied by loss of dermal white adipose tissue (DWAT), yet the mechanisms linking adipocyte depletion to fibroblast activation remain unclear. Here we identify the transcription factor SIX1 as a central regulator coupling adipogenic repression with profibrotic signaling. SIX1 expression was increased in skin biopsies from two independent SSc cohorts and localized to fibroblast and perivascular stromal cells. In mice, ubiquitous or adipocyte-specific deletion of Six1 preserved DWAT, reduced collagen accumulation, and selectively decreased pro-fibrotic mediators. In cultured fibroblasts, CRISPR/Cas9-mediated Six1 loss enhanced adipogenic markers while reducing profibrotic mediators and directly suppressed PAI-1 (SERPINE1) promoter activity. Together, these data position SIX1 as a transcriptional switch that promotes adipocyte reprogramming and fibrotic progression, and highlight SIX1 inhibition as a potential therapeutic strategy to preserve adipocyte identity and limit dermal fibrosis.
Authors
Nancy Wareing, Tingting W. Mills, Scott Collum, Minghua Wu, Lucy Revercomb, René Girard, Hui Liu, Alexes Daquinag, Mikhail Kolonin, Marka Lyons, Brian Skaug, Weizhen Bi, Meer A. Ali, Haniyeh Koochak, Anthony R. Flores, Yuntao Yang, W. Jim Zheng, William R. Swindell, Shervin Assassi, Harry Karmouty-Quintana
No posts were found with this tag.