Angiotensin signaling is essential for stress erythropoiesis but causes retention of dysfunctional mitochondria in RBCs
Rai et al. report that angiotensin signaling sustains erythroid expansion but also promotes retention of dysfunctional mitochondria in erythroid cells during stress erythropoiesis. The cover image is a collage of nucleated (purple) and enucleated cells from wild-type and ATR1-deficient mice. Mitochondria are highlighted in green. Image credit: Parul Rai and Madison Rice.
-
Research Letters
×
Abstract
Authors
Robert Lakin, Xueyan Liu, Dana Sherrard, Mihir Parikh, Ryan Debi, Nazari Polidovitch, Markus J. Duncan, Jian Wu, Peter H. Backx
Tiziana Parisi, Blanca Santibanez Ocampo, Jacob Adelman, Yuyan Cai, Marie McConkey, Christopher J. Gibson, Benjamin L. Ebert, Peter Miller, Tyler JacksTiziana Parisi, Blanca Santibanez Ocampo, Jacob Adelman, Yuyan Cai, Marie McConkey, Christopher J. Gibson, Benjamin L. Ebert, Peter Miller, Tyler Jacks×Abstract
Authors
Tiziana Parisi, Blanca Santibanez Ocampo, Jacob Adelman, Yuyan Cai, Marie McConkey, Christopher J. Gibson, Benjamin L. Ebert, Peter Miller, Tyler Jacks
-
Review
×
Abstract
The oxygen-rich milieu of the lungs necessitates precise iron homeostasis and regulation, processes that are fundamental to pulmonary physiology but often receive limited attention. However, in recent years, dysregulation of iron homeostasis has been linked to numerous acute and chronic respiratory diseases. Here, we comprehensively evaluate the mechanisms governing iron homeostasis in the alveolar epithelium of the lung and examine how iron dysregulation contributes to impaired alveolar epithelial repair in respiratory disease. This Review focuses on the effects of iron on alveolar epithelial cell homeostasis and repair and disease pathogenesis. There will be a focus on emerging interventions designed to reestablish iron homeostasis and their potential therapeutic implications related to enhancing lung repair and limiting the progression of lung disease.
Authors
Ugonna Mbaekwe, Sarah Kenny, Suzanne M. Cloonan, Corrine R. Kliment
-
Research Articles
Chiara Maria Assunta Cefalo, Teresa Mezza, Giuseppe Quero, Sergio Alfieri, Donatella Lucchetti, Filomena Colella, Alessandro Sgambato, Wei-Jun Qian, Andrea Mari, Alfredo Pontecorvi, Andrea Giaccari, Rohit N. KulkarniChiara Maria Assunta Cefalo, Teresa Mezza, Giuseppe Quero, Sergio Alfieri, Donatella Lucchetti, Filomena Colella, Alessandro Sgambato, Wei-Jun Qian, Andrea Mari, Alfredo Pontecorvi, Andrea Giaccari, Rohit N. Kulkarni×
Proteomic analyses of human islets reveal potential markers of β cell dysfunction during prediabetes
Abstract
The mechanisms driving progressive β cell dysfunction in type 2 diabetes remain incompletely understood. This study aimed to identify pancreatic islet proteome changes that could predict diabetes onset. We isolated islets from individuals without diabetes undergoing partial pancreatectomy, previously characterized for glucose tolerance, insulin sensitivity, and insulin secretion, using laser capture microdissection, and analyzed them via high-performance liquid chromatography–mass spectrometry. Proteomic analysis revealed that individuals with impaired glucose tolerance (IGT) had reductions in proteins regulating glycolysis (PGK1, G3P), lipid metabolism (ACBP, ARF1), glucose transport (14-3-3B), and insulin secretion (STARD10, CAPDS) compared with normal glucose-tolerant (NGT) individuals. Additionally, IGT islets showed impaired expression of proteins involved in glucose- and incretin-stimulated insulin response (CREB1, IQGA1). Stratification by β cell glucose sensitivity (βGS) indicated that individuals with lower βGS exhibited reduced levels of insulin maturation (ERO1B) and antiapoptotic proteins (CASP8, PAK2, SKP1), along with increased SEL1L, a factor promoting endocrine precursor differentiation. These findings suggest that early defects in glucose metabolism and insulin secretion characterize IGT, while reduced βGS may trigger compensatory mechanisms, through enhanced β cell survival or neogenesis, to delay type 2 diabetes progression. Overall, proteomic alterations in prediabetic islets provide potential early predictive markers and targets for interventions aimed at preserving β cell function.
Authors
Chiara Maria Assunta Cefalo, Teresa Mezza, Giuseppe Quero, Sergio Alfieri, Donatella Lucchetti, Filomena Colella, Alessandro Sgambato, Wei-Jun Qian, Andrea Mari, Alfredo Pontecorvi, Andrea Giaccari, Rohit N. Kulkarni
Toshiya Ichinose, Sho Tamai, Nozomi Hirai, Takashi Maejima, Kosuke Nambu, Hemragul Sabit, Shingo Tanaka, Masashi Kinoshita, Masahiko Kobayashi, Michihiro Mieda, Atsushi Hirao, Mitsutoshi NakadaToshiya Ichinose, Sho Tamai, Nozomi Hirai, Takashi Maejima, Kosuke Nambu, Hemragul Sabit, Shingo Tanaka, Masashi Kinoshita, Masahiko Kobayashi, Michihiro Mieda, Atsushi Hirao, Mitsutoshi Nakada×Abstract
Glioblastoma (GBM) is the most malignant primary brain tumor. The presence of glioma stem/initiating cells (GICs) is known to cause strong treatment resistance; therefore, GICs are a major target for GBM therapy, although there are no therapies targeting GICs clinically. To identify novel treatments for GBMs, we performed drug repurposing screening using GICs and identified the T-type calcium channel blocker lomerizine — a migraine prophylactic drug. Lomerizine inhibited proliferation, migration, invasion, and cell cycle progression and induced apoptosis in GICs and differentiated glioma cells. Lomerizine had antitumor effects by inactivating STAT3 in all cell lines. Furthermore, lomerizine also dephosphorylated AKT and ERK only in GICs and had strong tumor-suppressive ability. Lomerizine also reduced tumor volume and prolonged overall survival in vivo. Based on our data from in vitro and in vivo experiments, lomerizine has potential as a GBM therapeutic agent targeting both GICs and differentiated glioma cells and could benefit GBM patients.
Authors
Toshiya Ichinose, Sho Tamai, Nozomi Hirai, Takashi Maejima, Kosuke Nambu, Hemragul Sabit, Shingo Tanaka, Masashi Kinoshita, Masahiko Kobayashi, Michihiro Mieda, Atsushi Hirao, Mitsutoshi Nakada
Maile K. Hollinger, Chanie L. Howard, Donna C. Decker, Kelly M. Blaine, Ivy Aneas, Emily M. Grayson, Tania E. Velez, Fernando A. Oliveira, Riley T. Hannan, Daniel F. Camacho, Philip A. Verhoef, Cara L. Hrusch, Rebecca S. Griffes, Jeffrey M. Sturek, Marcelo A. Nobrega, Nathan Schoettler, Anne I. SperlingMaile K. Hollinger, Chanie L. Howard, Donna C. Decker, Kelly M. Blaine, Ivy Aneas, Emily M. Grayson, Tania E. Velez, Fernando A. Oliveira, Riley T. Hannan, Daniel F. Camacho, Philip A. Verhoef, Cara L. Hrusch, Rebecca S. Griffes, Jeffrey M. Sturek, Marcelo A. Nobrega, Nathan Schoettler, Anne I. Sperling×Abstract
Lung IL-33 is involved in pathogen defense, barrier homeostasis, and development of allergic responses. We previously identified a 5 kb noncoding region within a GWAS-defined segment that regulates expression of human IL33 (hIL33) but is absent in the murine locus. To understand how this region affects IL-33 expression in vivo, we engineered 2 BAC-transgenic strains in which 166 kb of the human genome upstream of the hIL33 locus, along with a fluorescent reporter, was inserted into the murine genome, both with and without the 5 kb region. Comparison to a murine Il33 (mIl33) reporter strain revealed species-specific tropism; hIL33 reporter was mostly expressed in the endothelium, while mIl33 reporter was expressed in type 2 alveolar epithelium. hIL33 reporter expression in tracheal basal epithelium, submucosal glands, and lung microvasculature required the 5 kb region. Surprisingly, allergen and exogenous IL-33 downregulated hIL33 reporter in lung endothelium only when the 5 kb region was present. Similar IL-33–dependent downregulation of IL33 transcripts was observed in human endothelial cell lines, validating that our hIL33 reporter strain recapitulated human endothelial biology. Together, these data reveal the importance of the asthma-associated human 5 kb region in regulating human IL33 expression in a cell type– and context-dependent manner.
Authors
Maile K. Hollinger, Chanie L. Howard, Donna C. Decker, Kelly M. Blaine, Ivy Aneas, Emily M. Grayson, Tania E. Velez, Fernando A. Oliveira, Riley T. Hannan, Daniel F. Camacho, Philip A. Verhoef, Cara L. Hrusch, Rebecca S. Griffes, Jeffrey M. Sturek, Marcelo A. Nobrega, Nathan Schoettler, Anne I. Sperling
×Abstract
T cells are the central players in antitumor immunity, and effective tumor killing depends on their ability to infiltrate into the tumor microenvironment (TME) while maintaining normal cytotoxicity. However, late-stage tumors develop immunosuppressive mechanisms that impede T cell movement and induce exhaustion. Investigating T cell migration in human tumors in vivo could provide insights into tumor immune escape, although it remains a challenging task. In this study, we developed ReMiTT, a computational method that leverages spatial transcriptomics data to track T cell migration patterns within tumor tissue. Applying ReMiTT to multiple tumor samples, we identified potential migration trails. On these trails, chemokines that promote T cell trafficking displayed an increasing trend. Additionally, we identified key genes and pathways enriched on these migration trails, including those involved in cytoskeleton rearrangement, leukocyte chemotaxis, cell adhesion, leukocyte migration, and extracellular matrix remodeling. Furthermore, we characterized the phenotypes of T cells along these trails, showing that the migrating T cells are highly proliferative. Our findings introduce an approach for studying T cell migration and interactions within the TME, offering valuable insights into tumor-immune dynamics.
Authors
Lin Zhong, Bo Li, Zhikai Chi, Siyuan Zhang, Qiwei Li, Guanghua Xiao
×Abstract
Sleep disturbance is a prevalent yet poorly understood comorbidity in autism spectrum disorders (ASD). Here, we uncover a bidirectional regulatory axis connecting the ASD risk gene POGZ to core circadian mechanisms. We demonstrate that Pogz is widely expressed in the suprachiasmatic nucleus (SCN), the central pacemaker of the circadian rhythms, and exhibits circadian oscillations in both the hypothalamus and liver, with its transcription directly regulated by the circadian molecule DBP through a D-box element in its proximal enhancer. Pogz-deficient mice exhibited prolonged circadian periodicity, impaired light-induced phase shift, delayed adaption to an 8-hour advance jet-lag, and reduced SCN c-Fos activation in response to light pulses. Mechanistically, POGZ interacts with and enhances the transcription activity of CREB, a key regulator of light-induced phase resetting. Notably, Pogz deletion leads to ASD-related deficits in social novelty and cognition, with cognitive impairments influenced by both photoperiod and behavioral paradigm. Our findings, thus, reveal a critical, previously unrecognized intersection between an ASD risk gene and circadian clock, offering insights into the pathogenesis of core ASD symptoms and comorbid sleep disturbances.
Authors
Ting Wu, Jiao He, Chu-Jun Xu, Chi-Yu Li, Pingchuan Zhang, Yanfeng Wang, Shanshan Zhu, Lusi Zhang, Jingtan Zhu, Jing Zhang, Jia-Da Li, Huadie Liu
×Abstract
Methyltransferase-like 5 (METTL5) is a methyltransferase responsible for rRNA N6-methyladenosine (m6A) modification, mutations in which are associated with skeletal abnormalities and cognitive deficits. Despite METTL5’s clinical relevance, the molecular mechanisms underlying METTL5-related genetic disorders remain poorly understood. In this study, we demonstrated that Mettl5 KO led to reduced bone mass and smaller body size in mice and impaired the osteogenic differentiation of mesenchymal stem cells. Mechanistically, Mettl5 deficiency decreased the translation efficiency of oxidative stress–responsive serine-rich protein 1 mRNA, downregulated the expression of key antioxidant genes, and diminished antioxidant capacity. Importantly, administration of the antioxidant N-acetylcysteine (NAC) partially rescued skeletal defects in Mettl5-KO mice. These findings reveal a critical role for METTL5 in antioxidant defense and suggest that NAC supplementation may represent a promising therapeutic strategy for METTL5-related disorders.
Authors
Kexin Lei, Qi Yin, Qiwen Li, Qian Wang, Zhong Zhang, Fei Xue, Ruoshi Xu, Xinyi Zhou, Lin Peng, Shoichiro Kokabu, Shuibin Lin, Quan Yuan
Zhenyuan Zhu, Mingjun Liu, Jianxin Wei, Deepa Suryanarayan, Parya Behzadi, Robert Edgar, Julie A. Phillippi, Cynthia St. Hilaire, Cristina Espinosa-Diez, Delphine GomezZhenyuan Zhu, Mingjun Liu, Jianxin Wei, Deepa Suryanarayan, Parya Behzadi, Robert Edgar, Julie A. Phillippi, Cynthia St. Hilaire, Cristina Espinosa-Diez, Delphine Gomez×Abstract
Thoracic aortic aneurysm and dissection (TAAD) involves a progressive dilation of the aortic wall associated with degradation of the extracellular matrix (ECM), cystic medial degeneration, smooth muscle cell (SMC) dysfunction, and rarefaction. TAAD etiology and pathogenesis suggest that alteration of mechanical force propagation may contribute to SMC dysfunction. This study aims to determine the role of SMC focal adhesion proteins, which are key components of force transmission, in TAAD pathogenesis. scRNA-seq analysis of human TAA aortas showed reduced expression of intracellular focal adhesion components, including PTK2 (FAK), VCL, ILK, and TES transcripts, in SMCs. Additionally, protein levels of FAK, ILK, and VCL were decreased in the aorta of patients with TAA. SMC-specific Ptk2, Vcl, and Ilk KO mice treated with β-aminopropionitrile (BAPN) exhibited increased mortality, aortic dilation, ECM breakdown, and SMC loss. Mechanistically, knocking down FAK, ILK, and VCL exacerbated gliotoxin-induced SMC anoikis, whereas overexpressing full-length WT and dead-kinase FAK conferred resistance to apoptosis and cell detachment, indicating that FAK’s protective effects depend on its expression rather than its enzymatic activity. Inhibition of FAK kinase activity did not affect SMC apoptosis in vitro or aortic dilation in vivo. Our findings demonstrate that the expression of focal adhesion proteins protects against TAAD progression and SMC anoikis independently of FAK kinase activity.
Authors
Zhenyuan Zhu, Mingjun Liu, Jianxin Wei, Deepa Suryanarayan, Parya Behzadi, Robert Edgar, Julie A. Phillippi, Cynthia St. Hilaire, Cristina Espinosa-Diez, Delphine Gomez
Galina Gritsina, Sandip Kumar Rath, Hongshun Shi, Qi Chu, Wanqing Xie, Que Thanh Waning Nguyen, Sambhavi Senthil, Thomas J. Myers, Mehmet A. Bilen, Sarah E. Fenton, Maha Hussain, David S. Yu, Jonathan C Zhao, Jindan YuGalina Gritsina, Sandip Kumar Rath, Hongshun Shi, Qi Chu, Wanqing Xie, Que Thanh Waning Nguyen, Sambhavi Senthil, Thomas J. Myers, Mehmet A. Bilen, Sarah E. Fenton, Maha Hussain, David S. Yu, Jonathan C Zhao, Jindan Yu×Abstract
VIC-1911 (formerly TAS-119) is a next-generation, ATP-competitive aurora kinase A (AURKA) inhibitor with a favorable biosafety profile. However, it has not been evaluated in prostate cancer (PCa), wherein AURKA is highly expressed in advanced stages and represents a critical therapeutic target. Here, we demonstrate that VIC-1911 potently inhibits AURKA activity with high selectivity over AURKB/C across diverse PCa cell lines. Treatment with VIC-1911, even at nanomolar concentrations, substantially inhibits the growth of both androgen receptor–positive (AR-positive) and AR-negative PCa cells. VIC-1911 triggers mitotic failure, induces DNA double-strand breaks (DSBs), and activates the p53 pathway, halting cell division and inducing cell death. Notably, VIC-1911 showed synergistic effects in inhibiting PCa cell growth in vitro and xenograft tumor growth in vivo with poly (ADP-ribose) polymerase inhibitors, which have proven effective in PCa with a deficiency in homologous recombination (HR) repair. Mechanistically, VIC-1911 disabled HR-mediated repair of DSBs in otherwise HR-proficient PCa cells, leading to a “BRCAness” phenotype and pronounced accumulation of DNA damage and mitotic catastrophe. In summary, our study uncovers what we believe is a novel mechanism to induce functional BRCAness through mitotic arrest and highlights VIC-1911 as a promising therapeutic agent for advanced PCa, either as a single agent or in combination, sensitizing HR-proficient tumors to PARP inhibitors.
Authors
Galina Gritsina, Sandip Kumar Rath, Hongshun Shi, Qi Chu, Wanqing Xie, Que Thanh Waning Nguyen, Sambhavi Senthil, Thomas J. Myers, Mehmet A. Bilen, Sarah E. Fenton, Maha Hussain, David S. Yu, Jonathan C Zhao, Jindan Yu
×Abstract
The molecular mechanisms responsible for the “atopic march” of allergic skin disease to allergic airway disease are incompletely understood. Secreted phospholipase A2 group X (sPLA2-X) is implicated in human asthma and modulates airway hyperresponsiveness (AHR) and inflammation in murine models of allergic asthma. We developed a complete proteolytic allergen model of dermal sensitization followed by airway challenge to mimic the “atopic march” and examined the role of sPLA2-X in regulating peripheral allergen sensitization, AHR, and airway inflammation. Pla2g10–/– mice receiving both house dust mite (HDM) peripheral sensitization and airway challenge had attenuated AHR relative to WT mice and lower airway eosinophils. Transgenic C57BL/6 hPLA2G10 mice (only expressing the human sPLA2-X gene) receiving treatment with a small molecule inhibitor of sPLA2-X (ROC0929) during the dermal sensitization phase demonstrated attenuated AHR and a reduction HDM-specific tissue-resident memory CD4+ T cells in the lung. Thus, sPLA2-X acts as an endogenous adjuvant to facilitate allergic sensitization in the periphery, which leads to AHR and airway inflammation following inhalation of the allergen. These results provide proof of concept that inhibition of sensitization in the periphery with a sPLA2-X inhibitor modulates subsequent allergen-induced airway dysfunction.
Authors
Ryan C. Murphy, Ying Lai, Yu-Hua Chow, Matt Liu, Brian D. Hondowicz, Dowon An, Marion Pepper, William A. Altemeier, Teal S. Hallstrand
×Abstract
N6-methyladenosine (m6A) modification is the most prevalent posttranscriptional epigenetic modification in mammalian mRNAs, and it has been implicated in the regulation of nervous system development by modulating mRNA metabolism. VIRMA is the largest core subunit of the m6A methyltransferase complex and is essential for the assembly and stability of the m6A methyltransferase complex. In the retina, m6A methylation modification is widely distributed in various cellular layers and is essential for retinal homeostasis. Here, we demonstrate that VIRMA-mediated m6A modification is essential for retinal homeostasis. Loss of Virma in retinal rod cells resulted in abnormal reduction in m6A methylation levels, along with impaired photoreceptor function and degeneration. Mechanically, Virma depletion in photoreceptors dampened the m6A modification level of visual perception–associated genes, resulting compromised visual function and photoreceptors degeneration. Moreover, Virma interacted with splicing factor to regulate the alternative splicing events of retina function–related genes such as Polg2, which contributes to photoreceptor damage. Reintroduction of normal Virma expression colonially rescued photoreceptor degeneration. Collectively, our data elucidate the important role of Virma-mediated m6A modification in photoreceptor function and suggest that epigenetic modulation could serve as a potential target to treat these blinding diseases.
Authors
Wenjing Liu, Xiaojing Wu, Rong Zou, Fan Zhang, Yudi Fan, Kuanxiang Sun, Liping Yang, Jiang Hu, Lin Zhang, Xianjun Zhu
Isaline Louvet, Ana Acosta-Montalvo, Chiara Saponaro, Maria Moreno-Lopez, Sana Douffi, Abdelkrim El Karchaoui, Gianni Pasquetti, Julien Thevenet, Nathalie Delalleau, Valery Gmyr, Paolo Giacobini, Stéphanie Espiard, Julie Kerr-Conte, François Pattou, Adrian Liston, Caroline BonnerIsaline Louvet, Ana Acosta-Montalvo, Chiara Saponaro, Maria Moreno-Lopez, Sana Douffi, Abdelkrim El Karchaoui, Gianni Pasquetti, Julien Thevenet, Nathalie Delalleau, Valery Gmyr, Paolo Giacobini, Stéphanie Espiard, Julie Kerr-Conte, François Pattou, Adrian Liston, Caroline Bonner×Abstract
HNF1A-MODY, the most common monogenic diabetes, exhibits progressive β cell dysfunction, but existing mouse models fail to recapitulate human disease progression, limiting understanding of pathogenic mechanisms. We developed mice with heterozygous deletion of the Hnf1a transactivation domain (Hnf1a+/Δe4-10) to model human HNF1A haploinsufficiency, conducted cross-sectional metabolic characterization, and validated our findings in HNF1A-deficient human islets. Unlike previous models, Hnf1a+/Δe4-10 mice successfully recapitulated temporal HNF1A-MODY progression. Male mice developed sequential pathophysiology: early insulin resistance in young adults (7 weeks), followed by testosterone deficiency and fasting hyperglycemia in adult mice (10 weeks). Glucose intolerance emerged in middle-aged mice (30 weeks), progressing to multi-organ dysfunction in aged mice (44–70 weeks), characterized by elevated hepatic gluconeogenesis, impaired renal glucose handling, and hepatic steatosis/fibrosis. This dual pathophysiology involving β cell dysfunction and peripheral insulin resistance was associated with dysregulated hormone secretion from both α and β cells in aged mice (40–70 weeks). Human islet studies with HNF1A knockdown confirmed translational relevance, demonstrating reduced SGLT2 protein expression and inappropriate glucagon and insulin secretion. This work established a physiologically relevant HNF1A-MODY model, identified early insulin resistance as a key mechanism triggering hormonal dysfunction, and revealed HNF1A’s role in multi-organ pathophysiology beyond traditional β cell dysfunction.
Authors
Isaline Louvet, Ana Acosta-Montalvo, Chiara Saponaro, Maria Moreno-Lopez, Sana Douffi, Abdelkrim El Karchaoui, Gianni Pasquetti, Julien Thevenet, Nathalie Delalleau, Valery Gmyr, Paolo Giacobini, Stéphanie Espiard, Julie Kerr-Conte, François Pattou, Adrian Liston, Caroline Bonner
×Abstract
A large interindividual variability in weight loss outcomes following bariatric surgery is reported. To ensure optimal management of patients, it is crucial to accurately identify candidates most likely to benefit the most from the intervention. Since genetic variants largely contribute to surgery response, polygenic scores (PGS) derived from genome-wide association studies (GWAS) could constitute valuable tools for clinical decision making. We developed and evaluated PGS to predict the weight loss response in 540 patients with a body mass index (BMI) of 35 kg/m2 or higher who underwent biliopancreatic diversion with duodenal switch. Summary statistics derived from BMI-derived GWAS, together with summary statistics from previously published GWAS of BMI and adiposity features, were used to construct, evaluate, and benchmark weight loss PGS. The full-adjusted BMI PGS model built in the entire cohort explained 39.6% of the mean-over-time excessive body weight loss (%EBWL), while the BMI-PGS built in the training dataset explained 38.9%. All benchmarked PGS based on BMI showed a significant relationship with mean-over-time %EBWL. These findings highlight the potential of BMI PGS in predicting weight loss after bariatric surgery and support their use as promising tools to improve the effectiveness of future antiobesity treatments.
Authors
Bastien Vallée Marcotte, Juan de Toro-Martín, André Tchernof, Louis Pérusse, Simon Marceau, Marie-Claude Vohl
Erin W. Kavanagh, Anya T. Joynt, Audrey R. Pion, Alice C. Eastman, Alianna I. Parr, Katherine L. Starego, Manav Jain, Sydney R. Shannon, Edwin J. Yoo, Gregory A. Newby, Stephany Y. Tzeng, Neeraj Sharma, Jordan J. Green, Garry R. CuttingErin W. Kavanagh, Anya T. Joynt, Audrey R. Pion, Alice C. Eastman, Alianna I. Parr, Katherine L. Starego, Manav Jain, Sydney R. Shannon, Edwin J. Yoo, Gregory A. Newby, Stephany Y. Tzeng, Neeraj Sharma, Jordan J. Green, Garry R. Cutting×Abstract
Cystic fibrosis (CF) is a life-limiting genetic disorder caused by deleterious variants in the CFTR gene that results in altered mucus impairing the airway epithelia. Durable correction of these variants in airway cells remains a therapeutic challenge for about 10% of individuals unresponsive to CFTR modulators. A common disease-causing CFTR splice site variant, 3120+1G>A, was corrected in primary CF airway cells using base editor RNAs. Single-cell RNA sequencing revealed a remarkable increase in detectable CFTR transcript in most CF airway epithelial cell types resulting in notable enrichment of CFTR-expressing ionocytes and secretory goblet cells. Progenitor basal cell subtypes were edited, but they decreased as a fraction of total cells and CFTR-expressing cells compared with unedited cells. CRISPR base editors delivered by polymeric nanoparticles (PNPs) facilitated functional rescue of CFTR to clinically meaningful levels in immortalized and primary airway cells. PNPs delivered GFP-encoding RNA to progenitor airway cells in fully differentiated airway cultures. Vitronectin was a major component of the PNP corona that formed in vivo, but preincubation with vitronectin did not enhance delivery. Together, these findings validate a scalable, nonviral platform with compelling translational promise for treating CF and other respiratory diseases involving respiratory epithelial cell dysfunction.
Authors
Erin W. Kavanagh, Anya T. Joynt, Audrey R. Pion, Alice C. Eastman, Alianna I. Parr, Katherine L. Starego, Manav Jain, Sydney R. Shannon, Edwin J. Yoo, Gregory A. Newby, Stephany Y. Tzeng, Neeraj Sharma, Jordan J. Green, Garry R. Cutting
×Abstract
Lysyl oxidase (LOX) is a copper-dependent monoamine oxidase whose primary function is the covalent cross-linking of collagen and elastin in the extracellular matrix (ECM). However, the regulation of LOX activity in renal fibrosis is not well understood. Here, our study showed that (a) LOX expression and ECM cross-linking were markedly increased in fibrotic kidneys. Reduction of copper levels in the Golgi apparatus by treatment with the copper chelator tetrathiomolybdate or by specific knockdown of copper transporter 1 (CTR1) decreased LOX activity and ameliorated renal fibrosis. (b) Overexpression of ATP7A caused an elevation of copper ions within the Golgi apparatus, resulting in increased LOX activity and enhanced ECM crosslinking, thereby promoting the progression of renal fibrosis. Knockdown of ATP7A showed the opposite result. (c) FBLN4 was essential for the ATP7A-mediated transfer of copper to LOX and formed a ternary complex of ATP7A-FBLN4-LOX. Our research revealed that high ATP7A expression induced copper overload in the Golgi apparatuses. FBLN4 then assisted ATP7A in transporting this excess copper to LOX, resulting in LOX overactivation. This, in turn, catalyzed the cross-linking of ECM components, thereby accelerating renal fibrosis.
Authors
Wenqian Zhou, Yan Zheng, Yuqing Liu, Jing Liu, Yiguo Liu, Yangyang Niu, Ying Yu, Xiaoqin Zhang, Yingying Zhang, Chen Yu
Heather E. True, Brianna M. Doratt, Sheridan B. Wagner, Delphine C. Malherbe, Nathan R. Shelman, Mahdi Eskandarian Boroujeni, Cynthia Cockerham, John M. O’Brien, Ilhem MessaoudiHeather E. True, Brianna M. Doratt, Sheridan B. Wagner, Delphine C. Malherbe, Nathan R. Shelman, Mahdi Eskandarian Boroujeni, Cynthia Cockerham, John M. O’Brien, Ilhem Messaoudi×Abstract
Maternal opioid use disorder (OUD) poses substantial risks to maternal and fetal health. These adverse outcomes are believed to be mediated, in part, by changes in placental structure and function; however, few studies have addressed this question. Here, we utilized flow cytometry, histology, and spatial and single-cell transcriptomics to uncover the impact of OUD on placental tissues. Given that half of individuals with chronic OUD contract hepatitis C (HCV), we further stratified our findings by maternal HCV status. Our results indicate that OUD leads to higher incidence of vascular malperfusion accompanied by increased levels of inflammatory markers and dysregulated secretion of placental development factors. Spatial transcriptomics revealed that OUD disrupts the communication between trophoblasts and immune cells important for placental vascular development. Additionally, CellChat analysis revealed aberrant vascular remodeling, neuropeptide, and chemotactic signaling across trophoblast, endothelial, and myeloid cells. Processes associated with tissue homeostasis and repair were also upregulated across trophoblasts and leukocytes. In addition, placental leukocytes were rewired toward regulatory/tissue surveillant phenotypes. Finally, frequencies and responses to ex vivo stimulation of decidual macrophages and cytolytic NK cells, critical for tissue remodeling and fetal tolerance, were decreased. Altogether, these results highlight substantial disruptions to placental health by maternal OUD.
Authors
Heather E. True, Brianna M. Doratt, Sheridan B. Wagner, Delphine C. Malherbe, Nathan R. Shelman, Mahdi Eskandarian Boroujeni, Cynthia Cockerham, John M. O’Brien, Ilhem Messaoudi
Mona Chatrizeh, Jianmin Tian, Matthew Rogers, Firuz Feturi, Guojun Wu, Brian Firek, Roman Nikonov, Lauren Cass, Alexandra Sheppeck, Lavnish Ojha, Ali Carroll, Matthew Henkel, Justin Azar, Rajesh K. Aneja, Brian Campfield, Dennis Simon, Michael J. MorowitzMona Chatrizeh, Jianmin Tian, Matthew Rogers, Firuz Feturi, Guojun Wu, Brian Firek, Roman Nikonov, Lauren Cass, Alexandra Sheppeck, Lavnish Ojha, Ali Carroll, Matthew Henkel, Justin Azar, Rajesh K. Aneja, Brian Campfield, Dennis Simon, Michael J. Morowitz×Abstract
Malnutrition, gut inflammation, and antibiotic-induced dysbiosis are well-recognized risk factors for poor clinical outcomes among critically ill patients. We previously showed that commercially available plant-based enteral nutrition (PBEN) preserves a commensal microbiome compared with commonly used artificial enteral nutrition (AEN). In this study, PBEN was superior to AEN in promoting recovery from antibiotic-induced dysbiosis in mice and humans. PBEN effectively mitigated anemia and leukopenia, restored naive lymphocyte populations, and reduced bone marrow myeloid expansion. Animals randomized to PBEN also exhibited improved responses to infectious gastrointestinal challenges following antibiotic exposure. A pilot clinical study validated these findings, demonstrating increased gut commensals, reduced pathogens, and improved leukocyte balance in critically ill children receiving PBEN compared with AEN. Together, these results suggest that PBEN offers a practical dietary approach to mitigate antibiotic-associated complications and potentially improve clinical outcomes among hospitalized patients requiring supplemental nutrition.
Authors
Mona Chatrizeh, Jianmin Tian, Matthew Rogers, Firuz Feturi, Guojun Wu, Brian Firek, Roman Nikonov, Lauren Cass, Alexandra Sheppeck, Lavnish Ojha, Ali Carroll, Matthew Henkel, Justin Azar, Rajesh K. Aneja, Brian Campfield, Dennis Simon, Michael J. Morowitz
Churaibhon Wisessaowapak, Yuliya Skorobogatko, Hyeonhui Kim, Xue Feng, Seunghwan Son, Haipeng Fu, Sitao Zhang, Pichaya Lertvilai, Lina Chang, Annie Hoang, Hetty Chen, Sarah Bedsted, Joseph Valentine, Jin Young Huh, Peng Zhao, Shannon M. Reilly, Piyajit Watcharasit, Maryam Ahmadian, Alan R. SaltielChuraibhon Wisessaowapak, Yuliya Skorobogatko, Hyeonhui Kim, Xue Feng, Seunghwan Son, Haipeng Fu, Sitao Zhang, Pichaya Lertvilai, Lina Chang, Annie Hoang, Hetty Chen, Sarah Bedsted, Joseph Valentine, Jin Young Huh, Peng Zhao, Shannon M. Reilly, Piyajit Watcharasit, Maryam Ahmadian, Alan R. Saltiel×Abstract
Metabolic adaptation to both caloric excess and restriction promotes energy conservation by suppressing catabolic pathways via feedback mechanisms that remain incompletely defined. We identified TANK binding kinase 1 (TBK1) as a nutrient- and inflammation-responsive brake on AMPK signaling in adipocytes. Fasting or pharmacological AMPK activation induced Tbk1 transcription via a PGC1α/nuclear respiratory factor 1 axis, which, in turn, limited AMPK activity through a phosphorylation cascade to conserve energy. In obesity, this AMPK/TBK1 axis was disrupted due to chronically elevated basal TBK1, thereby restricting energy expenditure during fasting. Adipocyte-specific TBK1 deletion enhanced fasting-induced AMPK activation, mitochondrial function, and lipolytic gene expression in both lean and obese mice. Pharmacological TBK1 inhibition with amlexanox recapitulated these effects. Combined treatment of mice with amlexanox and the AMPK activator AICAR enhanced weight loss, improved glucose tolerance and insulin sensitivity, and suppressed inflammatory and lipogenic programs in adipose tissue, as well as fibrotic gene expression in the liver. Building on prior clinical observations linking TBK1 inhibition to metabolic health, these findings defined a nutrient-sensitive AMPK/TBK1 feedback loop that limited adipocyte catabolism and suggested that dual targeting of TBK1 and AMPK may help counteract metabolic adaptation and enhance the durability of obesity therapies.
Authors
Churaibhon Wisessaowapak, Yuliya Skorobogatko, Hyeonhui Kim, Xue Feng, Seunghwan Son, Haipeng Fu, Sitao Zhang, Pichaya Lertvilai, Lina Chang, Annie Hoang, Hetty Chen, Sarah Bedsted, Joseph Valentine, Jin Young Huh, Peng Zhao, Shannon M. Reilly, Piyajit Watcharasit, Maryam Ahmadian, Alan R. Saltiel
×Abstract
Allogeneic islet transplantation is an effective treatment for type 1 diabetes, but its clinical use is limited by rejection involving innate and adaptive immune responses, requiring lifelong immunosuppression. We herein engineered islets that transiently display 2 immunomodulators chimeric with streptavidin (SA), thrombomodulin (SA-TM) and CD47 (SA-CD47), for localized modulation of both innate and adaptive immune responses. The engineering process did not impact islet viability, glucose responsiveness, and metabolic activity. Intraportal transplantation into allogeneic recipients achieved sustained survival, with 8 out of 11 grafts surviving 120–330 days without immunosuppression. In contrast, non-engineered islets were acutely rejected (median survival time [MST] = 12 days), while islets engineered with SA-TM showed delayed rejection (MST = 13.5 days) and those with SA-CD47 exhibited prolonged survival (MST = 24 days). Double-engineered islets generated a localized tolerogenic immune environment characterized by low frequencies of inflammatory innate immune cells and increased frequencies of M2 macrophages, myeloid-derived suppressor cells, and CD4+FoxP3+ T regulatory cells. The transcriptomic analysis showed downregulation of proinflammatory and upregulation of immune regulatory pathways. Our results demonstrate that transient co-display of immunomodulatory molecules on the islet surface is a versatile platform with significant translational potential for islet transplantation.
Authors
Shadab Kazmi, Mohammad Tarique, Darshan Badal, Vahap Ulker, Ali Turan, Kathleen M. Yee-Flores, Abdalmonam Jadou Nejma, Esma S. Yolcu, Haval Shirwan
Parul Rai, Swarnava Roy, Paritha Arumugam, Diamantis G. Konstantinidis, Sithara Raju Ponny, Marthe-Sandrine Eiymo Mwa Mpollo, Archana Shrestha, Theodosia A. Kalfa, Punam MalikParul Rai, Swarnava Roy, Paritha Arumugam, Diamantis G. Konstantinidis, Sithara Raju Ponny, Marthe-Sandrine Eiymo Mwa Mpollo, Archana Shrestha, Theodosia A. Kalfa, Punam Malik×Abstract
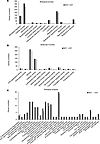
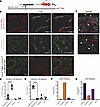
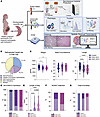
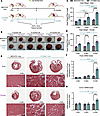
We previously reported that excessive angiotensin-II→AT receptor-1 (AT→ATR1) signaling results in sickle cell anemia–associated (SCA-associated) nephropathy. Herein, we showed that hyperangiotensinemia in SCA results from high erythroid cell–generated reactive oxygen species (ROS), which oxidized angiotensinogen (ATGN) and favored its rapid conversion to AT. Increased AT→ATR1 signaling in SCA erythroid cells generated ROS and created a positive feedback loop of ROS→oxidized ATGN→AT→ATR1→ROS, perpetuating the hyperangiotensinemia. ATR1 blocker, losartan, reduced erythrocyte ROS, oxidized ATGN, and AT levels. The ROS→AT→ATR1→ROS loop was driven by sickle erythropoiesis, as it was reproduced when WT mice were transplanted with SCA hematopoiesis. Using SCA and WT mice with germline- and erythroid-specific ATR1 deficiency, we found that stress erythropoiesis, but not steady-state erythropoiesis, was critically dependent on erythroid AT→ATR1 signaling, which acted in harmony with increased erythropoietin signaling. Furthermore, instead of the canonical AT→ATR1→NADPH-oxidase→ROS signaling in steady-state erythropoiesis, AT→ATR1 signaling in stress erythroid cells increased mitochondrial mass and dysfunctional mitochondria, which thereby increased ROS. SCA mice with erythroid-specific ATR1 deficiency had decreased RBC accumulation of dysfunctional mitochondria and decreased ROS, which reduced SCA-associated nephropathy. Overall, we demonstrate that AT→ATR1 signaling was essential for stress erythropoiesis but led to increased dysfunctional mitochondria retention in mature RBCs, which generated ROS and perpetuated hyperangiotensinemia, resulting in end-organ damage.
Authors
Parul Rai, Swarnava Roy, Paritha Arumugam, Diamantis G. Konstantinidis, Sithara Raju Ponny, Marthe-Sandrine Eiymo Mwa Mpollo, Archana Shrestha, Theodosia A. Kalfa, Punam Malik
Stephen B. Spurgin, Lauren Thai, Tina C. Wan, Christopher P. Chaney, Mitzy A. Cowdin, Surendranath Veeram Reddy, Tarique Hussain, Munes Fares, M. Luisa Iruela-Arispe, Thomas Carroll, Andrew D. Spearman, Ondine CleaverStephen B. Spurgin, Lauren Thai, Tina C. Wan, Christopher P. Chaney, Mitzy A. Cowdin, Surendranath Veeram Reddy, Tarique Hussain, Munes Fares, M. Luisa Iruela-Arispe, Thomas Carroll, Andrew D. Spearman, Ondine Cleaver×Abstract
Single-ventricle congenital heart disease (SV-CHD) is a uniformly lethal condition requiring the Glenn surgery, which as a side effect eliminates arterial pulsatility and contributes to pulmonary vascular complications. In Glenn patients, we quantified pulsatility loss in each dimension of force (flow, pressure, and stretch) using cardiac catheterization and MRI. To model and investigate the individual impact of each dimension of pulsatility loss on the pulmonary vasculature, we applied isolated pulsatile and non-pulsatile mechanical stimuli to pulmonary artery endothelial cells (ECs) in vitro. We found that each dimension of force triggered distinct transcriptional responses, revealing force-specific regulation of structural and signaling pathways. Pulsatile stretch uniquely stimulated EC secretion of PDGFB, a key driver of vascular smooth muscle cell (vSMC) recruitment. In a rat Glenn model, loss of pulsatility led to vascular wall thinning, loss of EC PDGFB, and reduced activation of smooth muscle PDGFBRβ, confirming in vivo relevance. Our findings uncover a mechanistic link between endothelial stretch sensing and PDGFB-mediated EC-vSMC crosstalk, essential for maintaining pulmonary artery architecture. Clinically, these insights suggest that restoring or mimicking pulsatile forces may help preserve vascular integrity and prevent remodeling in patients with SV-CHD.
Authors
Stephen B. Spurgin, Lauren Thai, Tina C. Wan, Christopher P. Chaney, Mitzy A. Cowdin, Surendranath Veeram Reddy, Tarique Hussain, Munes Fares, M. Luisa Iruela-Arispe, Thomas Carroll, Andrew D. Spearman, Ondine Cleaver
Takeshi Nakama, Aaron Wall, Garry Dolton, Li Rong Tan, Hannah Thomas, Hiroshi Hamana, Yoshiki Aritsu, Toong Seng Tan, Mako Toyoda, Yoshihiko Goto, Huanyu Li, Mizuki Kitamatsu, Keiko Udaka, Yusuke Miyashita, Hiroyuki Oshiumi, Kimitoshi Nakamura, Yoji Nagasaki, Rumi Minami, Hirotomo Nakata, Pierre J. Rizkallah, Hiroyuki Kishi, Takamasa Ueno, Andrew K. Sewell, Chihiro MotozonoTakeshi Nakama, Aaron Wall, Garry Dolton, Li Rong Tan, Hannah Thomas, Hiroshi Hamana, Yoshiki Aritsu, Toong Seng Tan, Mako Toyoda, Yoshihiko Goto, Huanyu Li, Mizuki Kitamatsu, Keiko Udaka, Yusuke Miyashita, Hiroyuki Oshiumi, Kimitoshi Nakamura, Yoji Nagasaki, Rumi Minami, Hirotomo Nakata, Pierre J. Rizkallah, Hiroyuki Kishi, Takamasa Ueno, Andrew K. Sewell, Chihiro Motozono×Abstract
Cytotoxic T lymphocytes form a critical component of SARS-CoV-2 immunity by recognizing viral peptides bound to HLA class I molecules. Here, we identified the spike-derived peptide NYNYLYRLF448-456 (NF9) as the immunodominant HLA-A*24:02–restricted epitope in both convalescent and vaccinated donors. Across cohorts, A24/NF9-specific responses were dominated by public TCR motifs featuring TRAV12-1 paired with TRBJ2-7 and a conserved CDR3β sequence (CASSXXXGYEQYF). Using a panel of 13 TCRs, we mapped recognition of single amino acid substitutions within NF9 and identified residue 5 (L452) as the principal determinant of escape. The L452R substitution, characteristic of the Delta variant, abolished recognition across all tested TCRs despite preserved HLA binding. Crystallography of a representative public TCR (P1-15) revealed that mutation at position-5 reoriented the peptide within HLA-A*24:02, flipping the adjacent Y453 side chain into the peptide-binding groove and eliminating the dominant TCR contact. This position-5–driven conformational switch provided a structural mechanism for universal loss of NF9 recognition by HLA-A*24:02–restricted T cells. Consistent with this, Delta-infected convalescents failed to mount de novo NF9-5R–specific responses while retaining responses to the conserved A24/QI9 spike epitope. Together, these findings defined the basis of A24/NF9 recognition and showed how 1 mutation remodeled peptide presentation to abrogate TCR responses.
Authors
Takeshi Nakama, Aaron Wall, Garry Dolton, Li Rong Tan, Hannah Thomas, Hiroshi Hamana, Yoshiki Aritsu, Toong Seng Tan, Mako Toyoda, Yoshihiko Goto, Huanyu Li, Mizuki Kitamatsu, Keiko Udaka, Yusuke Miyashita, Hiroyuki Oshiumi, Kimitoshi Nakamura, Yoji Nagasaki, Rumi Minami, Hirotomo Nakata, Pierre J. Rizkallah, Hiroyuki Kishi, Takamasa Ueno, Andrew K. Sewell, Chihiro Motozono
Declan L. Turner, Hannah Baric, Katelyn Patatsos, Sahel Amoozadeh, Michael See, Kathleen A. Strumila, Jack T. Murphy, Jeremy J. Wiyana, Liam Gubbels, Elizabeth S. Ng, Andrew G. Elefanty, Melanie R. Neeland, Shivanthan Shanthikumar, Sarah L. Londrigan, Mirana Ramialison, Fernando J. Rossello, Ed G. Stanley, Rhiannon B. WerderDeclan L. Turner, Hannah Baric, Katelyn Patatsos, Sahel Amoozadeh, Michael See, Kathleen A. Strumila, Jack T. Murphy, Jeremy J. Wiyana, Liam Gubbels, Elizabeth S. Ng, Andrew G. Elefanty, Melanie R. Neeland, Shivanthan Shanthikumar, Sarah L. Londrigan, Mirana Ramialison, Fernando J. Rossello, Ed G. Stanley, Rhiannon B. Werder×Abstract
The lung alveoli are continually exposed to inhaled pathogens and environmental hazards and rely on coordinated communication between alveolar macrophages and type 2 alveolar epithelial cells (AT2s) to maintain homeostasis. Disruption of these interactions can impair immunity and repair, contributing to acute and chronic respiratory diseases. To better define these mechanisms and support therapeutic discovery, we established a human iPSC-derived air-liquid interface platform that captures key features of AT2-macrophage crosstalk. Using this system, we show that coculture enhances AT2-specific transcriptional programs including lipid synthesis, while macrophages actively phagocytose AT2-derived surfactant. iPSC-derived macrophages adopt an alveolar macrophage–like phenotype and respond to AT2-derived M-CSF. During respiratory infection, macrophages play a crucial role in modulating epithelial inflammatory responses, augmenting antiviral immunity, and limiting viral replication. We further identify a role for macrophages in epithelial repair, where VEGF-mediated signaling to macrophages increases epithelial permeability during viral infection. Together, these findings reveal dimensions of AT2-macrophage cooperation in homeostasis, infection, and repair, and demonstrate how this iPSC-derived platform can be used to dissect mechanisms that may initiate or drive the progression of respiratory diseases.
Authors
Declan L. Turner, Hannah Baric, Katelyn Patatsos, Sahel Amoozadeh, Michael See, Kathleen A. Strumila, Jack T. Murphy, Jeremy J. Wiyana, Liam Gubbels, Elizabeth S. Ng, Andrew G. Elefanty, Melanie R. Neeland, Shivanthan Shanthikumar, Sarah L. Londrigan, Mirana Ramialison, Fernando J. Rossello, Ed G. Stanley, Rhiannon B. Werder
Dilraj Kaur, Sagarika Chakrabarty, Claudius Witzler, Hongjie Wang, Mengwen Wang, Romina Wolz, Petra Wilgenbus, Jens J.N. Posma, Sivaramakrishna Rachakonda, Federico Marini, Valeriya V. Zinina, Sabine Reyda, Rajinikanth Gogiraju, Claudine Graf, Fahumiya Samad, Katrin Schäfer, Christoph Reinhardt, Natalia Soshnikova, Wolfram Ruf, Thati MadhusudhanDilraj Kaur, Sagarika Chakrabarty, Claudius Witzler, Hongjie Wang, Mengwen Wang, Romina Wolz, Petra Wilgenbus, Jens J.N. Posma, Sivaramakrishna Rachakonda, Federico Marini, Valeriya V. Zinina, Sabine Reyda, Rajinikanth Gogiraju, Claudine Graf, Fahumiya Samad, Katrin Schäfer, Christoph Reinhardt, Natalia Soshnikova, Wolfram Ruf, Thati Madhusudhan×Abstract
Glucagon-like peptide-1 (GLP-1) and glucose-induced insulinotropic polypeptide (GIP) receptor agonists have revolutionized obesity therapy, but causes of obesity-associated dysregulation of endogenous incretin production remain incompletely understood. Here we show that intestinal transmembrane serine protease 2 (TMPRSS2) plays a pivotal role in deregulating anti-diabetic GLP-1 production in obesity. TMPRSS2 is widely coexpressed in intestinal epithelial cells along with its signaling target protease-activated receptor 2 (PAR2). In addition to its role in regulating coagulation protease–mediated adipose tissue inflammation, PAR2 signaling in the gut controls postprandial GIP secretion. TMPRSS2, but not the epithelial cell–expressed proteases FXa or matriptase, activates PAR2 and thereby promotes postprandial GIP release. Accordingly, a PAR2-mutant mouse resistant to TMPRSS2 cleavage is protected from GIP upregulation and diet-induced obesity. In the context of obesity, TMPRSS2 also attenuates bioavailability of the ghrelin pathway and thereby suppresses GLP-1–mediated control of glucose homeostasis. Pharmacological inhibition or genetic deletion of TMPRSS2 restores ghrelin signaling–dependent GLP-1 secretion and GLP-1’s anti-diabetic effects on nutritional glucose homeostasis. Thus, epithelial cell–expressed TMPRSS2, which critically contributes to the lung pathology in SARS-CoV-2 infection, emerges as an intestinal incretin regulator and a potential link between infection and chronic cardiometabolic diseases.
Authors
Dilraj Kaur, Sagarika Chakrabarty, Claudius Witzler, Hongjie Wang, Mengwen Wang, Romina Wolz, Petra Wilgenbus, Jens J.N. Posma, Sivaramakrishna Rachakonda, Federico Marini, Valeriya V. Zinina, Sabine Reyda, Rajinikanth Gogiraju, Claudine Graf, Fahumiya Samad, Katrin Schäfer, Christoph Reinhardt, Natalia Soshnikova, Wolfram Ruf, Thati Madhusudhan
×Abstract
Myotonic dystrophy type 1 (DM1) is caused by an expanded CTG repeat in the DMPK gene, resulting in mutant transcripts that form expanded CUG (CUGexp) RNA foci and sequester muscleblind-like (MBNL) RNA-binding proteins. DM1 is multisystemic, with progressive worsening of disease manifestations in affected tissues. Disease progression is attributed to somatic expansion of the CTG repeats with age, resulting in production of CUGexp RNA with enhanced intrinsic toxicity due to increased MBNL sequestration. To determine the degree to which cardiac disease progression can occur independently of repeat expansion, we used a transgenic DM1 mouse model with inducible heart-specific expression of a stable, interrupted 960-CUG-repeat RNA. Sustained CUGexp RNA expression caused progressive cardiac enlargement, contractile dysfunction, conduction delay, myocardial fibrosis, and reduced survival, while MBNL-dependent splicing defects remained static, consistent with the stable repeat length. We also determined the degree of reversibility after different periods of CUGexp RNA expression by shutting off the repeat-containing transgene. Suppression of CUGexp RNA expression rescued cardiac abnormalities, but reversibility declined with longer exposure to the toxic RNA. These findings demonstrate that prolonged expression of stable CUGexp RNA drives progressive cardiac pathology, revealing a mechanism of disease progression in DM1 in addition to somatic expansion.
Authors
Rong-Chi Hu, Mohammadreza Tabary, Xander H.T. Wehrens, Thomas A. Cooper
Raquel Gómez-Oca, Xènia Massana-Muñoz, David Reiss, Juliana De Carvalho Neves, Nadege Diedhiou, Roberto Silva-Rojas, Belinda S. Cowling, Marie Goret, Jocelyn LaporteRaquel Gómez-Oca, Xènia Massana-Muñoz, David Reiss, Juliana De Carvalho Neves, Nadege Diedhiou, Roberto Silva-Rojas, Belinda S. Cowling, Marie Goret, Jocelyn Laporte×Abstract
Centronuclear myopathies (CNMs) are rare congenital disorders characterized by muscle weakness, fiber hypotrophy, and organelle mislocalization. Most cases arise from mutations in MTM1 or DNM2, encoding myotubularin and dynamin-2, respectively. DNM2 is a GTPase that binds lipids, oligomerizes around membranes, and mediates fission. We previously showed that DNM2 levels are elevated in MTM1-CNM patients and Mtm1–/y mice, and that normalizing DNM2 rescues disease phenotypes. However, the specific DNM2 functions driving pathology remain unclear. Here, we expressed AAV-delivered WT and DNM2 mutants in WT and Mtm1–/y mouse muscles to disrupt specific DNM2 molecular functions. In WT mice, overexpression of WT DNM2 and most mutants induced CNM-like phenotypes, including reduced force, fiber hypotrophy, and centralized nuclei, consistent with gain-of-function mechanisms. The lipid-binding-defective mutant K562E did not induce disease-like phenotype. In Mtm1–/y mice, K562E mutant markedly improved muscle force, mass, and fiber size, while others failed to rescue. Therefore, we generated Mtm1–/y Dnm2K562E/+ mice, which showed full rescue of survival, motor function, and muscle force, with improved muscle mass, fiber size, and organelle positioning despite persistently elevated DNM2 levels. This study reveals that DNM2 lipid binding, not protein abundance or GTPase activity, drives pathology, and represents the most rational therapeutic target for DNM2 therapy in MTM1-CNM.
Authors
Raquel Gómez-Oca, Xènia Massana-Muñoz, David Reiss, Juliana De Carvalho Neves, Nadege Diedhiou, Roberto Silva-Rojas, Belinda S. Cowling, Marie Goret, Jocelyn Laporte